Microfluidic Droplet Generation Technology Guide: Principles, Wafer Devices and Applications
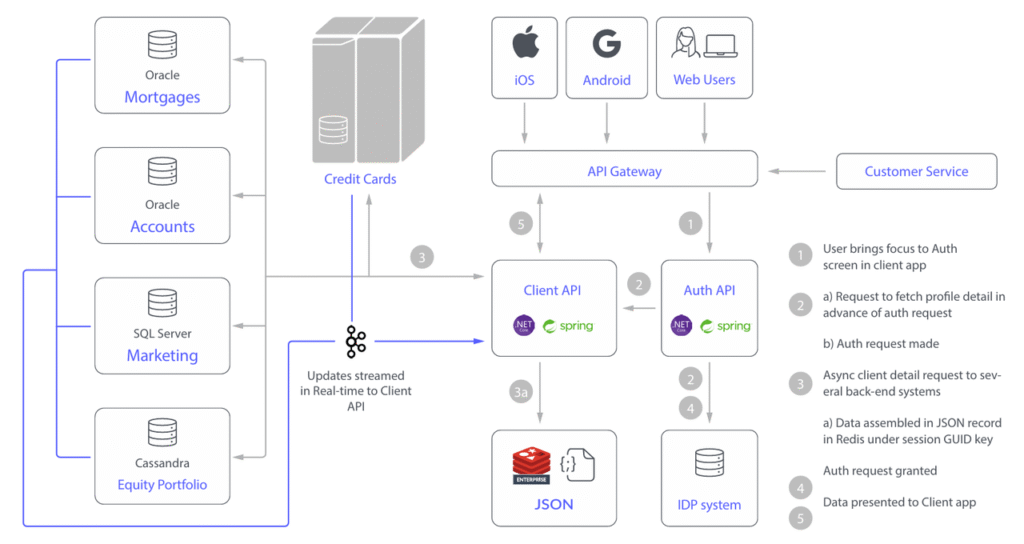
Looking for microfluidic solutions for high-throughput single-cell analysis and drug discovery? HONGKE provides you with the analysis of microfluidic droplet generation principles (T-junction, fluidic focusing), high-precision wafers and pressure pumping equipment to help you optimize your experimental process. Read more!