Hongke Solutions] Decision Automation vs Process Automation: Breaking the Bottleneck of B2B Digital Transformation
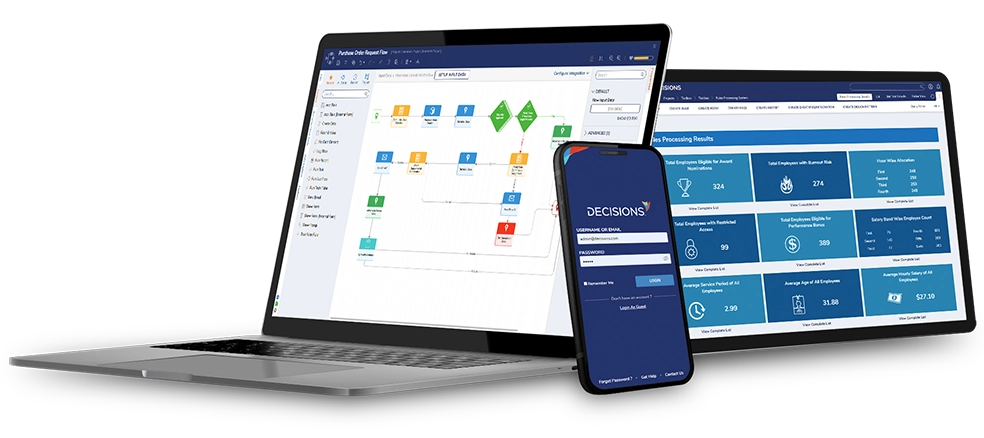
Just because a process is fast doesn't mean a decision is fast! HongKe explains how to systematize "decision logic" through the Decisions platform. From discount authorization to risk management, decision automation can dramatically improve consistency and compliance, helping Hong Kong and Southeast Asian companies achieve high-quality growth. Learn more about our automation upgrades today!



